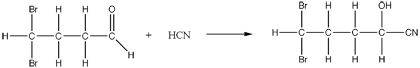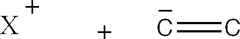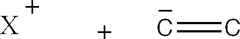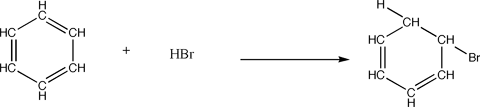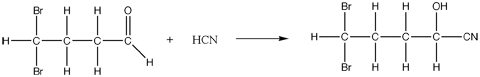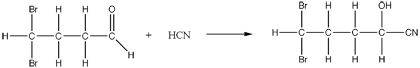Go Back Home

Electrophilic Addition Reactions and Mechanisms
Characteristics:
- The reactant must contain at least one carbon to carbon double or triple bond. In other words,the reactant must be an alkene or alkyne group.
What Actually Happens
- A pi bond is broken by the formation of two new covalent bonds.
- The driving force for this reaction is the formation of 'X' ans an electophile which forms a covalent bond with one of the double or triple bonded carbons.
- Because 'X' is and electrophile, it has a positive charge which is then tranfered to one of the carbons within the double or triple bond.
- The positive charge is then transferred to the other carbon within the double or triple bond.
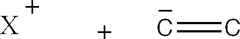
The positively charged carbon intermediate combines with electron-rich 'Z' to form the second covalent bond.

Examples!


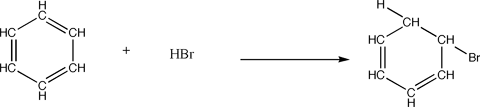
Nucleophilic Addition Reactions and Mechanisms
Characteristics
- Usually between a carbonyl compound (typically an aldehyde or ketone) and hydrogen cyanide.
What Actually Happens
- Hydrogen cyanide adds across the carbon to carbon double bond, breaking the pi bond and forming two covalent bonds.

- The nucleophilic cyanide attacks the slightly positive carbon atom.
- The slightly negative ion then picks up a hydrogen ion from the hydrogen cyanide ion.
Ketones:
- Ketones undergo nucleophilic addition reactions in much the same way as aldehydes:
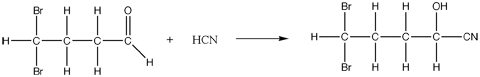
Note: optical isomerism also becomes important for a compound that has four different groups attached to a central carbon atom. For the sake of simplicity, this web page will not cover this topic.
Examples!